Capa report8/16/2023 
There is much to be learned from the manufacturing sector when it comes to leveraging CAPA, not only for compliance purposes but to help accelerate timeĮssential CAPA principles come from regulations and quality standards. 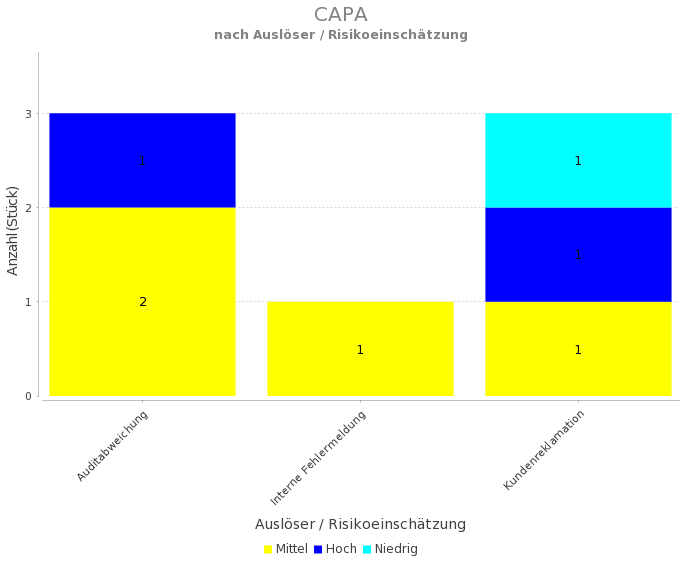
Indeed the term “clinical quality management system” or CQMS was coined by MasterControl only five years ago. But, unlike in manufacturing, there’s less emphasis in formalizing CAPA procedures and embedding them in a clinical trial’s quality process, or in integrating them with the QMS. So why can’t clinical research professionals adopt CAPA in clinical trials? In fact, they have been using CAPA more than most people realize. Companies regulated by the US Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other regulatory entities, as well as companies that adhere to ISO quality standards, ICH quality guidelines, and similar international standards, typically maintain a CAPA process as part of their quality management system (QMS). If the issue is significant, it will be escalated into a corrective action and preventative action (CAPA).ĬAPA is inherently tied to the concept of product quality in manufacturing. 
If the problem is minor and there’s a solution that can be performed immediately and sufficiently, then the quality event can be closed with an effective containment or correction. When that happens, the quality issue must be resolved in a timely, effective and compliant manner. In pharmaceutical and biopharmaceutical manufacturing, it is generally assumed that somewhere along the process of manufacturing a product, something is likely to go wrong that could affect quality. The diagram shows the CAPA process, from start to finish.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
